
The coverage on this live blog has ended — but for up-to-the-minute coverage on the coronavirus, visit the live blog from CNBC's U.S. team.
Stocks are again set to rise on promising Covid vaccine news. Pfizer announced early Wednesday that final data analysis shows its vaccine candidate is 95% effective — better than the previously reported effectiveness of "more than 90%." The company also said the drug worked consistently across all ages, races and ethnicities. Dow Jones Industrial Average futures rose in early trading Wednesday.
Here are some of today's biggest developments:
- Pfizer says final analysis shows the vaccine is 95% effective
- Moderna's vaccine maker details 'big challenges' in mass production
- Gottlieb says Covid stimulus, sacrifice needed for possibly 'the last time'
- Vaccine shots could go to health workers in about a month
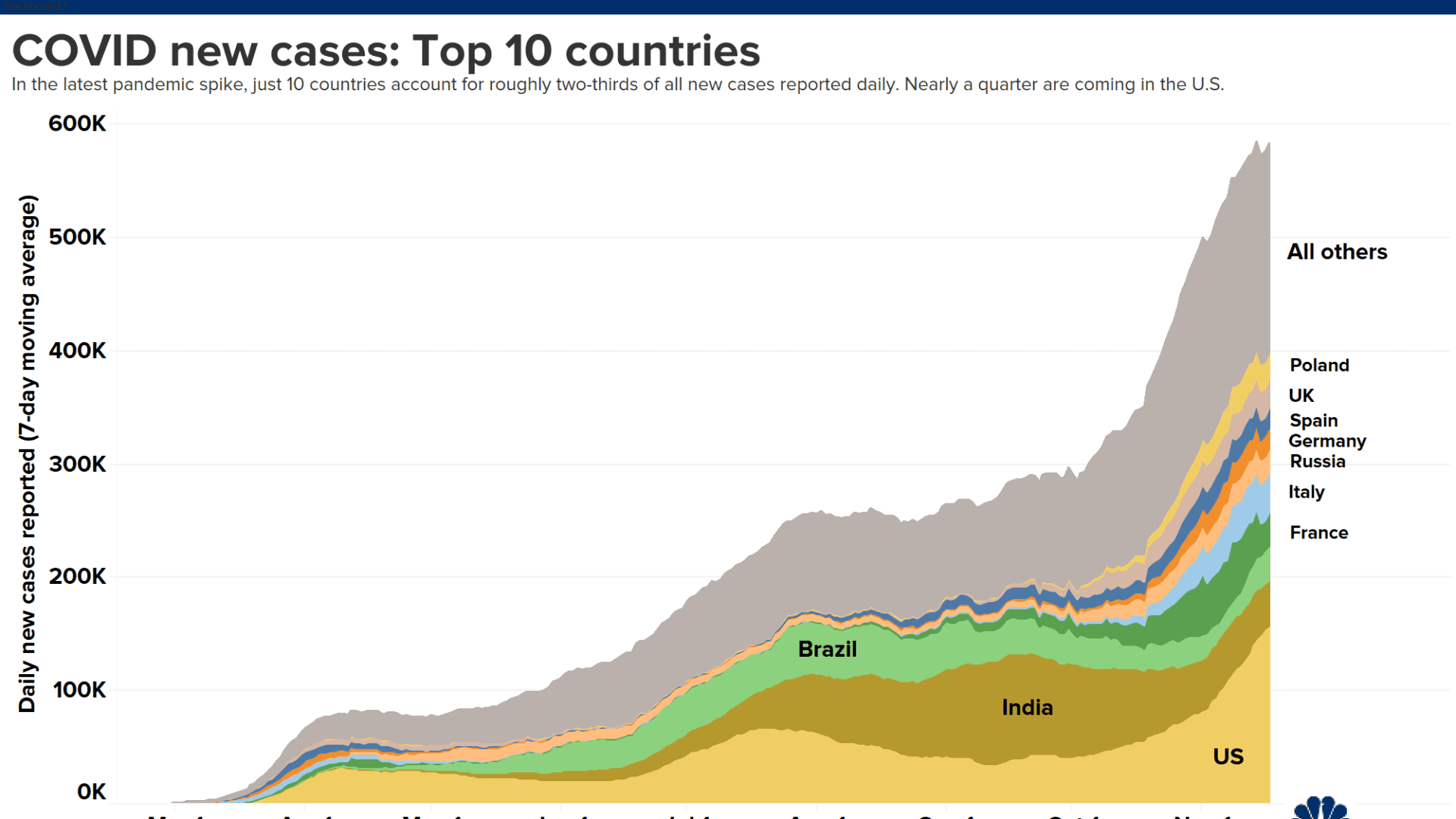
The following data was compiled by Johns Hopkins University:
- Global cases: More than 56.2 million
- Global deaths: At least 1.34 million
- U.S. cases: More than 11.52 million
- U.S. deaths: At least 250,485
Money Report
Generic drugmakers in India will be "essential" in manufacturing and distributing Covid-19 therapeutics to low- and middle-income countries, according to United Nations-backed Medicines Patent Pool.
The public health body told CNBC that most of the drugmakers it is already working with to expedite access to coronavirus treatments are based within the South Asian country.
India is home to the world's largest generic drugs market and has previously proven itself instrumental in distributing low-cost medicines, particularly to poorer nations.
"There could be a really important role for Indian generic drugmakers," executive director Charles Gore said of the fight to combat the coronavirus which has so far infected more than 56.1 million people — 8.9 million in India alone.
—Karen Gilchrist
The NFL announced that every team will be under strict Covid-19 safety protocols beginning Saturday as the U.S. struggles with slowing a spike in cases and hospitalizations, the Associated Press reports.
The new restrictions apply to every level of team activities, including:
- Mandatory mask-wearing at all times in team facilities, including during practice and in weight rooms.
- Meetings held virtually or in the largest indoor space with the league's approval.
- Meals available for grab-and-go to avoid large numbers of people in one area.
- Locker rooms will also be limited to avoid large crowds.
"The upcoming holidays, beginning with Thanksgiving next week, will introduce new risks of exposure that we need to address now," NFL Commissioner Roger Goodell wrote in a note to teams.
—Chris Eudaily
As the coronavirus seeps into nearly every corner of the U.S., a handful of states and cities are closing nonessential businesses, limiting public and private gatherings and imposing mask mandates to try to slow the rapid spread ahead of the winter season.
Without a cohesive federal plan, officials are reinstituting some of the stay-at-home orders, curfews and other public safety measures that were rolled back over the summer and fall. Many governors and mayors have made it clear, however, that they don't want to shut down the economy again like many did in March and April at the outset of the outbreak.
A handful of Democrat governors in Washington, Oregon and New Mexico have imposed more sweeping measures, banning indoor dining and placing limits on gatherings. Some Republican leaders in Iowa, North Dakota and Utah, many of whom have long resisted statewide lockdowns and mask requirements, are now ordering residents to wear face coverings in public.
"Masks do not negatively affect our economy and wearing them is the easiest way to slow the spread of the virus," Utah Gov. Gary Herbert said during a
—Noah Higgins-Dunn
The United States is in the midst of an "absolutely dangerous situation" as the coronavirus outbreak continues to worsen, assistant secretary of health Adm. Brett Giroir said.
"Right now, we are in an absolutely dangerous situation that we have to take with the utmost seriousness," he said on MSNBC. "This is not crying wolf. This is the worst rate of rise in cases that we've seen in the pandemic in the United States and right now there's no sign of flattening."
Giroir's comments come after the latest weekly report issued by the White House coronavirus task force, which was obtained Tuesday by NBC News, warned of "aggressive, unrelenting, expanding broad community spread across the country, reaching most counties, without evidence of improvement but rather, further deterioration."
"We all know what the data show. The data are absolutely concerning," Giroir said. "I lose sleep at night over where we are in the pandemic right now, but we all also know how to beat this."
—Will Feuer
New York City's decision to close schools for in-person learning beginning Thursday is being criticized by some.
The move had been anticipated for days after de Blasio told parents on Friday to have a plan in place in case the city decided to close schools for in-person learning. The mayor said the city would close classrooms once the citywide positivity rate, or the percent of Covid-19 tests that are positive, hits an average of 3%.
"We set a very clear standard and we need to stick to that standard, and I want to emphasize to parents, to educators, to staff, to kids, that we intend to come back and come back as quickly as possible," de Blasio said during a press briefing.
Some, however, have criticized the decision to close schools as gyms and indoor dining at restaurants remain open at reduced capacity.
"THIS IS TOTALLY BACKWARDS," Mark Levine, a New York City councilmember who chairs the health committee, said in a tweet posted after the announcement.
Dr. Celine Gounder, an infectious disease specialist at the New York University Grossman School of Medicine and a coronavirus advisor to President-elect Joe Biden, told CNBC last week that she was concerned the city's "prioritizing the wrong things" as the mayor instructed families to prepare for online learning.
"The problem is not coming from the schools. It's coming from the bars, the restaurants, the gyms and the living room family spread," Cuomo said on a call with reporters last week. "So if in fact you do close schools, I would urge the mayor and all involved to open them as quickly as possible."
— Noah Higgins-Dunn
Sen. Elizabeth Warren is calling on the Centers for Disease Control and Prevention to clarify and revise its estimates for the risk posed to communities of color by Covid-19.
"By failing to adjust COVID-19 mortality rates by age in its public data releases, the CDC may not be providing an accurate assessment of the increased risk of death and serious illness for communities of color relative to white Americans of the same age," the Massachusetts Democrat said last week in a letter to CDC Director Dr. Robert Redfield.
"The fact that the average age among communities of color is much younger than that of nonHispanic white Americans makes the disproportionate number of deaths among communities of color all the more disturbing," she wrote. "To date, the CDC has not consistently articulated risks and has not provided full and complete information on the degree to which age and race or ethnicity interact to inflate the risk of COVID-19 mortality for communities of color."
—Will Feuer
Google employee productivity is nearly back to pre-pandemic levels thanks to a combination of leadership coaching and employee surveys, CFO Ruth Porat said.
Speaking at Wednesday's New York Times DealBook conference, Porat said Google experienced a dip in employee productivity — particularly in junior levels— in the early days of the pandemic.
She said that the company realized it needed to double down on coaching senior leadership to train those under then for times of crisis.
Porat also said Google has conducted many employee surveys, which it checked against data it had on coding to assess how productive people were being. She said planning employees' return to offices next year has been "substantially more complicated" than it was when preparing employees to work from home full-time.
—Jennifer Elias
The Trump administration is seeing high uptake of a program that will allow CVS Health and Walgreens to administer Covid vaccines to seniors in long-term care facilities, according to Health and Human Services Secretary Alex Azar said.
He said 99% of skilled nursing facilities across the country have signed up for the program, which will given Covid-19 vaccines to seniors free of charge. Facilities in at least 20 states are fully signed up. "The ultimate goal here is to make getting a Covid-19 vaccine as convenient as getting a flu shot," he told reporters during a press conference.
The Trump administration first announced the program with CVS and Walgreens in October. The program is optional, and the facilities can opt-in to the program through the CDC's National Healthcare Safety Network.
–Berkeley Lovelace Jr.

The New York City school system will temporarily close on Thursday, Mayor Bill de Blasio announced.
"We're in the middle of something really tough right now," de Blasio said at a press briefing Monday. "We have put health and safety first, and we will put health and safety first."
The shuttering of the nation's largest school system had been anticipated for days after de Blasio told parents on Friday to have a plan in place in case the city decides to close schools for in-person learning, NBC News New York reported. The shutdown was prompted after the city reached a 3% testing positivity rate over a seven-day average.
—Riya Bhattacharjee, Noah Higgins Dunn
Dr. Mike Ryan, executive director of the World Health Organization's health emergencies program, said that a coronavirus vaccine this winter won't help countries beat back the current wave of Covid-19 infections.
"We're not there with vaccines yet. We will get there, but we're not there," Ryan said during a social Q&A event. "And many countries are going through this wave, and they're going to go through this wave and continue through this wave without vaccines."
The outcomes of severely-ill patients could also grow worse in some countries with overburdened health systems since health-care workers are forced to ration their time and attention, he said.
"Not only do we have full hospitals, but in those hospitals, patients won't do so well," he said. "There's only one way to stop more people from needing to go to the hospital, and that's to stop more people getting exposed and getting infected."
—Noah Higgins-Dunn
U.S. health-care workers, the first in line for a coronavirus vaccine once authorized by the Food and Drug Administration, could get their shots in about a month.
Pfizer announced that it planned to submit an application to the FDA for emergency use "within days" after a final data analysis found its vaccine was highly effective in preventing Covid-19. The FDA process is expected to take a few weeks, and an advisory committee meeting to review the vaccine has been tentatively scheduled for early December.
The vaccine is expected to be released in phases, with health-care workers and vulnerable Americans, such as the elderly and those with underlying health conditions, getting it first. Essential workers, teachers and people in homeless shelters, as well as people in prisons, would likely be next on the list. That's followed by children and young adults.
—Berkeley Lovelace Jr.

Delta Air Lines is sticking with its policy of blocking middle seats until March 30, extending a policy the airline has used during the pandemic to stand out among rivals.
Most of Delta's competitors have already dropped the policy, or plan to sell all seats on board in the coming weeks. Delta had previously said it would limit capacity on board through at least Jan. 6.
Airlines are competing for precious few passengers as most would-be travelers continue to avoid air travel. Delta and other carriers say their on-board filtration systems make airplane cabins a highly unlikely place to catch the coronavirus.
The Atlanta-based carrier's chief customer officer Bill Lentsch said: "We recognize some customers are still learning to live with this virus and desire extra space for their peace of mind. We are listening and will always take the appropriate steps to ensure our customers have complete confidence in their travel with us."
—Leslie Josephs

Pfizer and Moderna have released promising late-stage clinical trial data showing their coronavirus vaccines, both using a new messenger RNA technology, were highly effective in preventing Covid-19.
Now, the challenge will be figuring out how to distribute their vaccines, both of which require two shots, to billions of people across the globe. It will likely take months, maybe even more than a year, to distribute enough doses for the U.S. and the rest of the world to suppress the virus, public health officials and health experts warn.
In the U.S., Health and Human Services Secretary Alex Azar told CNBC on Monday that there will be roughly 40 million doses of vaccine available by the end of this year between the two companies, enough to inoculate about 20 million people.
"Still, this process could be much more complicated and more chaotic than people think," said Yanzhong Huang, a senior fellow for global health at the Council on Foreign Relations, adding 40 million doses in 2020 will not be sufficient to achieve so-called herd immunity in the U.S.
—Noah Higgins-Dunn, Berkeley Lovelace Jr.
Former Food and Drug Administration chief Dr. Scott Gottlieb called on Americans to take steps to reduce coronavirus spread, while also urging Congress to provide fiscal support to aid businesses such as bars and restaurants that may be ordered to close as the crisis worsens.
"This may be the last time we have to do it, so we really should do what we can to try to preserve life over the next two or three months, recognizing that there's really a better future ahead of us, with respect at least to Covid-19 in 2021," Gottlieb said on CNBC's "Squawk Box."
The reason for optimism, Gottlieb said, is due to the positive developments around Covid-19 vaccines, with those from Pfizer and Moderna both indicating efficacy of around 95%. "We see the light at the end of the tunnel now," said Gottlieb, a Pfizer board member. "These vaccines could effectively end the ... U.S. epidemic in 2021 as we more widely deploy them."
—Kevin Stankiewicz
A group of advisors has been asked by the Food and Drug Administration to set aside three days in early December for potential meetings to discuss Covid-19 vaccines, CNBC's Meg Tirrell reports, citing two people familiar with the plans. The meetings would be a key step in the agency's emergency authorization process.
The group may be asked to comment on Pfizer and Moderna's vaccines, said the sources, who asked not to be named.
The meetings, said to be set for Dec. 8, 9 and 10, would come just weeks after both companies reported data from their phase three trials showing their vaccines are about 95% effective in preventing cases of Covid-19.
—Terri Cullen

Target said it won market share across all of its core categories during the third quarter as pandemic shopping habits proved long-lasting.
Sales rose 20.7% year over year, bolstered by a 155% rise in comparable digital sales.
Target locations remained open in the early days of virus shutdowns, and the company beefed up delivery options to reach Americans stuck at home. The company said Wednesday it held onto those March and April customers and won more of their shopping spend, CNBC's Melissa Repko reports.
Sales were higher in every category: Electronics sales shot up by more than 50%, the home items category rose by a mid-20s percentage, apparel jumped by nearly 10%, and essentials & beauty and food & beverage each grew in the high teens.
—Sara Salinas
Moderna's announcement earlier this week that its vaccine was more than 94% effective at preventing the coronavirus, according to preliminary trial data, raised global hopes further that an end to the pandemic that has killed over 1.3 million people could be in sight.
But mass producing a vaccine at speed, overseeing that manufacturing process, and then transporting it around the world pose "big challenges," according to Swiss drugmaker Lonza that has partnered with Moderna to produce its coronavirus vaccine.
"We can only produce more than 500 million doses a year if we install additional manufacturing lines, so it is clear that we need additional investments in installation if we want to produce more than 500 million (per year) in the future," Lonza Chairman Albert Baehny told CNBC's "Squawk Box Europe."
—Holly Ellyatt
Pfizer said a final data analysis found its coronavirus vaccine was 95% effective in preventing Covid-19, was well tolerated and appeared to fend off severe disease.
The company, which has been developing the vaccine with German drugmaker BioNTech, said it plans to submit an application for emergency use authorization to the FDA "within days." Pfizer CEO Albert Bourla said at Tuesday's New York Times Dealbook conference that the company had accumulated enough safety data needed to submit the vaccine for review.
The news comes more than a week after the companies announced that their vaccine was more than 90% effective and two days after Moderna said preliminary phase three trial data showed its vaccine was 94.5%. Both vaccines use messenger RNA, or mRNA, technology.
–Berkeley Lovelace Jr.
The U.S. is recording roughly 157,000 new Covid cases per day, on average, as of Tuesday, according to a CNBC analysis of Johns Hopkins data. That's another new record and nearly 30% higher than infection levels a week ago.
The national seven-day average of new cases has been at record levels — and rising — since Oct. 25, with infections and current hospitalizations now spiking in nearly every state, according to CNBC's analysis.
—Sara Salinas






